Walgreens Pain Reliever Acetaminophen, 150 count bottles
Class II - ModerateWhat Should You Do?
- Check if you have this product: (About 137,300)
- Do not eat it: Even if it looks and smells fine, do not consume this product.
- Throw it away or return it: You can return the product to the store for a full refund.
- Report problems: Report any issues to the FDA's Safety Reporting Portal.
Recall Details
- Company:
- Aurobindo Pharma Limited, of Telangana, India
- Reason for Recall:
- The recalled over-the-counter products contain the regulated substance acetaminophen which must be in child resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging of the products is not child resistant, posing a risk of poisoning if the contents are swallowed by young children.
- Classification:
- Class II - Moderate
Products that might cause a temporary health problem, or pose a slight threat of a serious nature.
- Status:
- ongoing
- Hazard:
- The recalled over-the-counter products contain the regulated substance acetaminophen which must be in child resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging of the products is not child resistant, posing a risk of poisoning if the contents are swallowed by young children.
- Remedy:
- Consumers should immediately store the recalled products in a safe location out of reach and sight of children. Contact Aurohealth for information on how to return the product at your nearest Walgreens store to receive a full refund.
Product Images
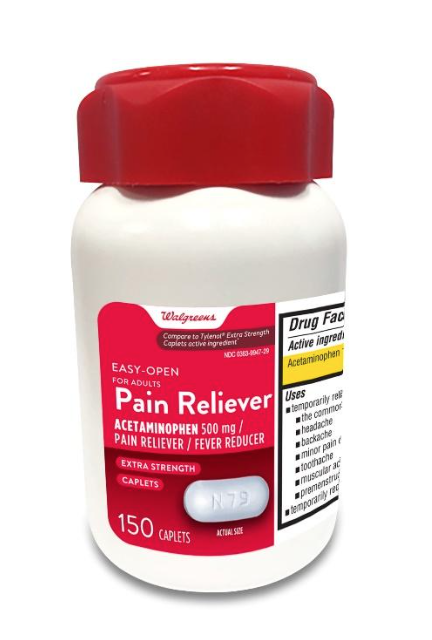

Product Information
Full Description:
This recall involves the Walgreens brand acetaminophen product. The red and white label states Walgreens, Easy Open for Adults, Pain Reliever, Acetaminophen, 500 mg, Fever Reducer, Extra Strength, 150 caplets. The bottle has a red continuous thread gear closure. UPC number 311917218090 and Lot numbers P2100627, P2100671, P2100672, P2100689 P2100747, P2100859 (each with expiration date Nov-2022) and P2200050 (with expiration date Jan-2023) are included in this recall. The UPC number, lot numbers and expiration date are printed near the drug facts panel on the label on the back of the bottle.
Product Codes/Lot Numbers:
(About 137,300)
Official Source
Always verify recall information with the official CPSC source:
View on CPSC.govCPSC Recall Number: 22161
Related Recalls
Kroger Arthritis Pain Acetaminophen, 225 count bottles
Aurobindo Pharma Limited, of Telangana, India
The recalled over-the-counter product contains the regulated substance acetaminophen which must be in child resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging of the products is not child resistant, posing a risk of poisoning if the contents are swallowed by young children.